UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: +
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange | ||
| Item 7.01 | Regulation FD Disclosure. |
From time to time, Wave Life Sciences Ltd. (the “Company”) presents and/or distributes slides and presentations to the investment community to provide updates and summaries of its business. On October 2, 2023, the Company updated its corporate presentation, which is available on the “For Investors & Media” section of the Company’s website at http://ir.wavelifesciences.com/. This presentation is also furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The information in this Item 7.01 and exhibit 99.1attached hereto is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed incorporated by reference into any registration statement or other filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 9.01 | Financial Statements and Exhibits. |
| (d) | Exhibits. |
The following exhibit relating to Item 7.01 is furnished and not filed:
| Exhibit No. | Description | |
| 99.1 | Corporate Presentation of Wave Life Sciences Ltd. dated October 2, 2023 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| WAVE LIFE SCIENCES LTD. | ||
| By: | /s/ Paul B. Bolno, M.D. | |
| Paul B. Bolno, M.D. | ||
| President and Chief Executive Officer | ||
Date: October 2, 2023

Exhibit 99.1 Wave Life Sciences Corporate Presentation October 2, 2023

Forward-looking statements This document contains forward-looking statements. All statements other than statements of historical facts contained in this document, including statements regarding possible or assumed future results of operations, preclinical and clinical studies, business strategies, research and development plans, collaborations and partnerships, regulatory activities and timing thereof, competitive position, potential growth opportunities, use of proceeds and the effects of competition are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause the actual results, performance or achievements of Wave Life Sciences Ltd. (the “Company”) to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “aim,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this presentation are only predictions. The Company has based these forward-looking statements largely on its current expectations and projections about future events and financial trends that it believes may affect the Company’s business, financial condition and results of operations. These forward-looking statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties and assumptions, including those listed under Risk Factors in the Company’s Form 10-K and other filings with the SEC, some of which cannot be predicted or quantified and some of which are beyond the Company’s control. The events and circumstances reflected in the Company’s forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Moreover, the Company operates in a dynamic industry and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that the Company may face. Except as required by applicable law, the Company does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. 2

Wave today is well positioned for significant and sustained growth DMD, HD, and AATD clinical programs advancing Collaborations & access to genetic insights DMD, HD, and AATD clinical programs advancing Clinical proof-of- Leader in RNA editing therapeutics, emerging concept leader in RNAi Opened new Multi-modal drug discovery biology and development platform Platform Pipeline of novel medicines for rare and learnings prevalent diseases Data Science Strategic collaborations to Foundation expand and advance pipeline Chemistry innovation GMP manufacturing Stereochemistry Strong and broad IP Building a leading RNA medicines company 3

RNA medicines allow matching disease target to therapeutic modality Most versatile RNA medicines Restored platform (PRISM™) in the Reading Frame industry Endogenous ADAR enzyme Functional Best-in-class nucleic acid Protein chemistry applicable editing splicing across modalities Ability to access novel / untapped areas of disease biology Platform learnings and clinical validation continue RISC to increase probability Endogenous Endogenous of success AGO2 RNase H silencing silencing 4
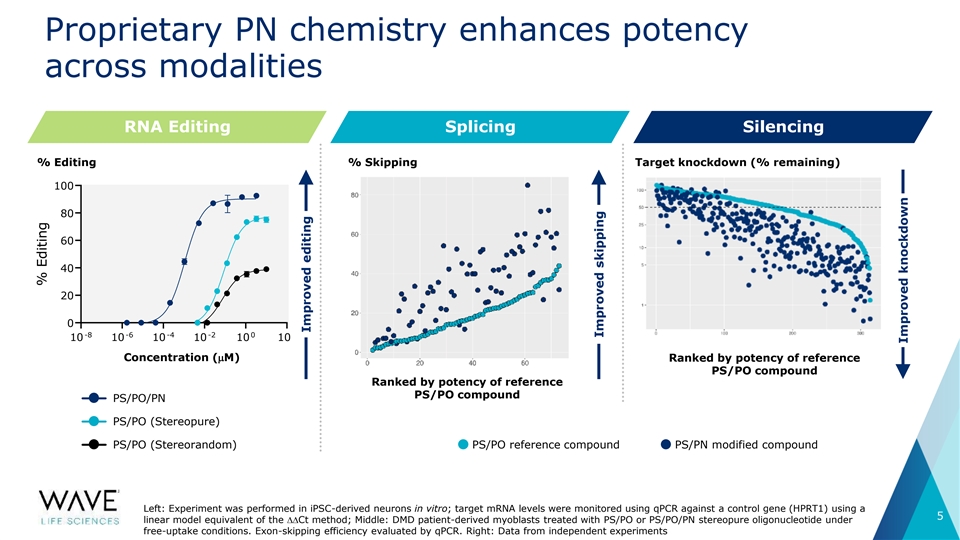
Proprietary PN chemistry enhances potency across modalities RNA Editing Splicing Silencing % Editing % Skipping Target knockdown (% remaining) 100 80 60 40 20 0 -8 -6 -4 -2 0 2 10 10 10 10 10 10 Concentration (µM) Concentration (mM) Ranked by potency of reference PS/PO compound Ranked by potency of reference PS/PO compound PS/PO/PN PS/PO (Stereopure) PS/PO (Stereorandom) PS/PO reference compound PS/PN modified compound Left: Experiment was performed in iPSC-derived neurons in vitro; target mRNA levels were monitored using qPCR against a control gene (HPRT1) using a 5 linear model equivalent of the DDCt method; Middle: DMD patient-derived myoblasts treated with PS/PO or PS/PO/PN stereopure oligonucleotide under free-uptake conditions. Exon-skipping efficiency evaluated by qPCR. Right: Data from independent experiments % Editing Improved editing Improved skipping Improved knockdown

Robust RNA medicines pipeline including first-in-class RNA editing programs Patient population Program Discovery Preclinical Clinical Rights (US & Europe) RNA EDITING WVE-006 GSK exclusive 200K SERPINA1 (AATD) global license Multiple undisclosed 100% global >20K (multiple) Correction Multiple undisclosed 100% global >3M (multiple) Upregulation SPLICING WVE-N531 Phase 1/2 100% global 2.3K Exon 53 (DMD) Other exons (DMD) 100% global Up to 18K SILENCING: ANTISENSE Takeda 50:50 25K Manifest (SNP3) WVE-003 Phase 1/2 mHTT (HD) Option 60K Pre-Manifest (SNP3) SILENCING: RNAi INHBE* (Metabolic disorders, including 100% global 47M obesity) *Through GSK collaboration, Wave can advance up to three collaboration programs (the first of which is INHBE) and GSK can advance up to eight collaboration programs. 6 AATD: Alpha-1 antitrypsin deficiency; DMD: Duchenne muscular dystrophy; HD: Huntington’s disease Editing for correction Editing for upregulation
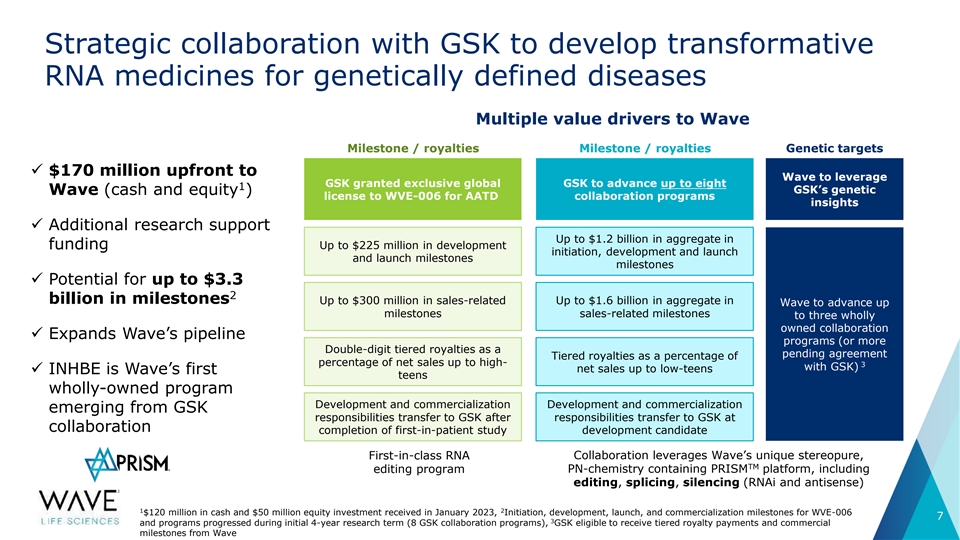
Strategic collaboration with GSK to develop transformative RNA medicines for genetically defined diseases Multiple value drivers to Wave Milestone / royalties Milestone / royalties Genetic targets ✓ $170 million upfront to Wave to leverage GSK granted exclusive global GSK to advance up to eight 1 GSK’s genetic Wave (cash and equity ) license to WVE-006 for AATD collaboration programs insights ✓ Additional research support Up to $1.2 billion in aggregate in funding Up to $225 million in development initiation, development and launch and launch milestones milestones ✓ Potential for up to $3.3 2 billion in milestones Up to $300 million in sales-related Up to $1.6 billion in aggregate in Wave to advance up milestones sales-related milestones to three wholly owned collaboration ✓ Expands Wave’s pipeline programs (or more Double-digit tiered royalties as a pending agreement Tiered royalties as a percentage of percentage of net sales up to high- 3 with GSK) net sales up to low-teens ✓ INHBE is Wave’s first teens wholly-owned program Development and commercialization Development and commercialization emerging from GSK responsibilities transfer to GSK after responsibilities transfer to GSK at collaboration completion of first-in-patient study development candidate First-in-class RNA Collaboration leverages Wave’s unique stereopure, TM editing program PN-chemistry containing PRISM platform, including editing, splicing, silencing (RNAi and antisense) 1 2 $120 million in cash and $50 million equity investment received in January 2023, Initiation, development, launch, and commercialization milestones for WVE-006 7 3 and programs progressed during initial 4-year research term (8 GSK collaboration programs), GSK eligible to receive tiered royalty payments and commercial milestones from Wave

WVE-N531 Duchenne muscular dystrophy

Duchenne muscular dystrophy • Genetic mutation in dystrophin gene Disease State Restored State prevents the production of dystrophin Dysfunctional Splicing Exon Skipping Oligo protein, a critical component of healthy Mutant pre-mRNA Mutant pre-mRNA muscle function 50 51 53 54 55 50 51 53 54 55 • Impacts approx. 1 in every 5,000 Skip newborn boys each year; approx. 20,000 new cases annually worldwide 50 51 53 54 55 50 51 54 55 – Approx. 8-10% are amenable to exon 53 skipping mRNA with disrupted reading frame Restored mRNA • Dystrophin protein established by FDA as Translation halted Translation continues surrogate endpoint reasonably likely to 1 predict benefit in boys for accelerated approval in DMD • Increasing amount of functional dystrophin expression over minimal amount shown with approved therapies is expected to result No dystrophin Functional in greater benefit for boys with DMD protein produced dystrophin produced 1 9 Vyondys: www.fda.gov; viltepso; www.fda.gov; Exondys; www.fda.gov; Amondys: www.fda.gov

Extended survival in dKO preclinical model supports potential of exon-skipping therapeutics for DMD PN chemistry improved function and survival in dKO mice dKO survival studies in literature Restored muscle and respiratory 100% survival at time of study termination function to wild-type levels 300 200 100 0 20 40 60 80 100 120 Stimulation Frequency (Hz) Wild-type dKO: PBS dKO: PS/PO/PN Tidal volume Time (weeks) PS/PO/PN 150 mg/kg weekly PS/PO/PN 75 mg/kg bi-weekly PS/PO 150 mg/kg weekly PBS Age (days) Wild-type dKO: PBS dKO (PS/PO/PN Note: Untreated, age-matched mdx mice had 100% survival at oligonucleotide) study termination [not shown] 10 Left: Kandasamy et al., 2022; doi: 10.1093/nar/gkac018; Right: Forand et al., 2020; doi: https://doi.org/10.1016/j.omtm.2020.03.011. Survival probability (%) 2 TVb (ml) Specific Force (Nm )
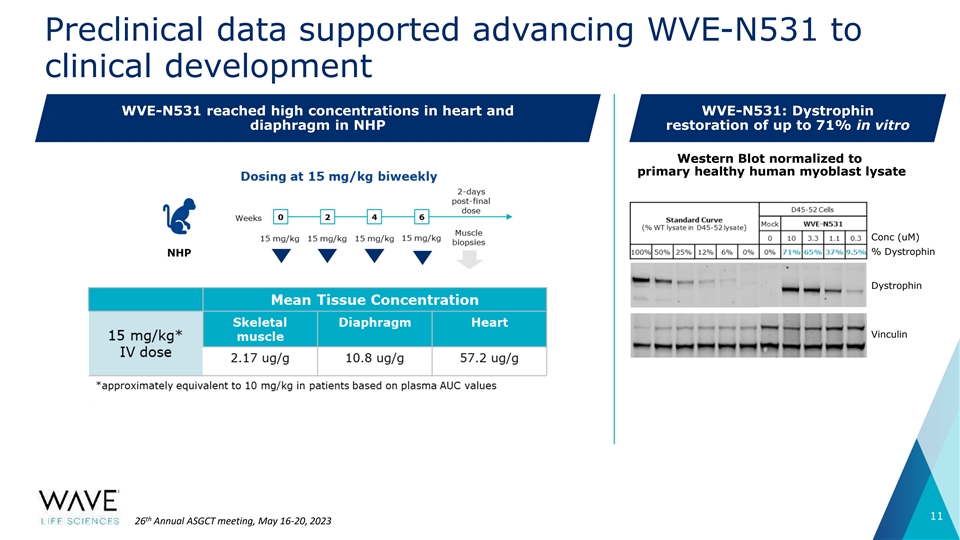
Preclinical data supported advancing WVE-N531 to clinical development WVE-N531 reached high concentrations in heart and WVE-N531: Dystrophin diaphragm in NHP restoration of up to 71% in vitro Western Blot normalized to primary healthy human myoblast lysate Conc (uM) % Dystrophin Dystrophin Vinculin 11 th 26 Annual ASGCT meeting, May 16-20, 2023

WVE-N531 Part A clinical data: High exon-skipping & muscle concentrations after three bi-weekly doses WVE-N531 uptake in WVE-N531 uptake in myocyte stem cells myocyte stem cells suvodirsen WVE-N531 Mean muscle 0.7 µg/g 42 µg/g concentration Mean exon Not detectable 53% skipping WVE-N531 uptake in Half-life in myogenic stem 18 hours 25 days cells plasma 22 weekly doses of 5 3 biweekly doses of 10 Dose mg/kg mg/kg Important for potential muscle regeneration WVE-N531 data presented March 22, 2023 at Muscular Dystrophy Association Clinical and Scientific Conference; WVE-N531 biopsies collected ~2 weeks post-last dose (3 biweekly 12 doses of 10 mg/kg) 42 µg/g = 6.1 µM; Suvodirsen biopsies collected post-last dose (weekly doses of 5 mg/kg) on week 22; Half-life as indicated by PK analysis; suvodirsen: discontinued first-generation non-PN chemistry compound; Right: Dual staining utilizing in-situ hybridization for WVE-N531 and PAX7 immunohistochemistry for stem cells

FORWARD-53, a potentially registrational Phase 2 clinical trial of WVE-N531 in DMD (Exon 53) Screening Biweekly Dosing (10 mg/kg IV) Safety Follow-up • Functional • Biopsy after 24 weeks of • Biopsy after 48 weeks of treatment assessment treatment • Functional assessment • Functional assessment • Design of FORWARD-53: Phase 2, open-label, 10 mg/kg every other week, up to 10 patients • Endpoints: Dystrophin (powered for >5% of normal), safety/tolerability, pharmacokinetics, functional assessments (incl. NSAA and others) • Biopsies: – After 24 weeks of treatment – After 48 weeks of treatment Data from FORWARD-53 expected in 2024 13 IV: intravenous; NSAA: North star ambulatory assessment

Potential for Wave to address up to ~40% of DMD population Exon skipping and dystrophin restoration demonstrated in vitro Exon Skipping 1.1 µM 2.5 µM 2.5 µM DMD Population 3.3 µM 5 µM 5 µM 100 100 100 10 µM 10 µM 10 µM WVE-N531 Exon 53 Not Amenable 50 50 50 to Skipping 8-10% Exon 51 17% 0 0 0 11-13% Ex51 Ex51 mock Ex52 Ex52 mock Ex44 Ex44 mock SSO-1 SSO-2 SSO-1 SSO-2 SSO-1 SSO-2 6% Exon 44 Protein Restoration 4% 1.1 µM 2.5 µM 2.5 µM 150 150 Exon 52 3.3 µM 5 µM 5 µM 100 10 µM 10 µM 10 µM 8% Exon 45 100 100 44% 50 Other Exons 50 50 0 0 0 Ex51 Ex51 mock Ex52 Ex52 mock Ex44 Ex44 mock SSO-1 SSO-2 SSO-1 SSO-2 SSO-1 SSO-2 Exon 51 Exon 52 Exon 44 14 Left: Aartsma-Rus, et al. 2009 Hum Mutat 30, 293. % Dystrophin Relative to WT % Exon 51 Skipping Rel to Total DMD % Dystrophin Relative to WT % Exon 52 Skipping Rel to Total DMD % Dystrophin Relative to WT % Exon 44 Skipping Rel to Total DMD

WVE-006 for Alpha-1 antitrypsin deficiency (AATD)

WVE-006: Designed to correct mutant SERPINA1 transcript to address both liver and lung manifestations of AATD WVE-006 designed to correct WVE-006 ADAR editing approach to address key goals of AATD treatment: Z allele mRNA to enable M-AAT protein to be produced 2) Reduce Z-AAT 1) Restore circulating, 3) Retain M-AAT protein aggregation in A functional wild-type M-AAT physiological regulation liver SERPINA1 Z allele mRNA encodes Z-AAT protein with E342K mutation Z-AAT WVE-006 (GalNAc- conjugated AIMer) I(G) RNA correction replaces M-AAT reaches lungs to M-AAT secretion into mutant Z-AAT protein protect from proteases bloodstream with wild-type M-AAT protein Edited SERPINA1 mRNA enables wild- type M-AAT protein production 200,000 Pi*ZZ patients in US and Europe AAT: Alpha-1 antitrypsin Strnad et al., 2020 N Engl J Med 382:1443-55; Blanco et al., 2017 Int J Chron Obstruct Pulmon Dis 12:561-69; Remih 16 et al., 2021 Curr Opin Pharmacol 59:149-56.

WVE-006 in AATD: First-in-class RNA editing clinical candidate Potentially comprehensive approach to address both lung and liver manifestations of AATD Increased AAT protein Confirmed restored Demonstrated functionality in NSG-PiZ mice wild-type M-AAT protein of M-AAT protein WVE-006 treatment results in serum AAT Overall percentages of serum AAT Serum neutrophil elastase protein levels of up to 30 uM in NSG-PiZ mice protein isoforms in NSG-PiZ mice inhibition activity in NSG-PiZ mice (Week 13) 2000 PBS 1800 WVE-006 1600 WVE-006 (NO LOADING DOSE) 1400 1200 ~7-fold 1000 increase 800 600 11μM 400 200 0 Week ~50% editing supports restoration of MZ phenotype AATD: Alpha-1 antitrypsin deficiency; M-AAT protein: wild-type AAT protein; WVE-006 administered subcutaneously (10 mg/kg bi-weekly) in 7-week old NSG-PiZ mice (n=5 per group); Loading dose: 3 x 10 mg/kg at Day 0. Left: Liver biopsies collected at wk 13 (1 wk after last dose) and SERPINA1 editing quantified by Sanger sequencing; Right: Total 17 serum AAT protein quantified by ELISA; Stats: Two-Way ANOVA with adjustment for multiple comparisons (Tukey) 0 1 2 3 4 5 6 7 8 9 10 11 12 13 Serum AAT protein (ug/ml) (Mean, s.e.m)

WVE-006 decreases lobular inflammation and PAS-D globule size, prevents increase in hepatocyte turnover Fibrosis → Cirrhosis → Hepatocellular Carcinoma Correction of gain-of-function liver phenotypes Lobular inflammation Mitoses PAS-D-positive globule size (NSG PiZ mice, week 13) (NSG PiZ mice, week 13) (NSG PiZ mice, week 13) ns✱✱✱✱ ns ✱✱✱✱ 5 40 ns ✱✱✱ 25✱✱✱✱ ✱ 4 20 30 3 15 20 2 10 10 5 1 0 0 0 Week 0 Week 13 Week 0 Week 13 Week 0 Week 13 Left (Lobular inflammation) and Middle (Mitoses): Scatter plot showing inflammation grade or mitoses score. Each circle represents an individual mouse, (Mean ± SEM); Right (PAS-D Globule Size): 40 largest globules in each of 5 mice were measured. Each circle represents a single PAS-D globule, (Mean ± 18 SEM). Baseline: week 0 (7 weeks old); Treated week 13 (20 weeks old); Stats: Kruskal-Wallis followed by Dunn’s test Baseline PBS WVE-006 Baseline PBS WVE-006 Baseline PBS WVE-006 Score (0-4) Number of mitotic figures/10 MPF + Mean PAS-D globule diameter (μm)

AIMer-directed editing is highly specific in mice No bystander editing observed on SERPINA1 transcript RNA editing only detected at PiZ mutation site in SERPINA1 transcript RNA editing across transcriptome (mouse liver) (mouse liver) C 0% SERPINA1 PBS (PiZ mutation site) T 100% C 48.2% AATD AIMer T 51.8% % Editing Editing site (PiZ mutation) Dose 3x10 mg/kg (days 0, 2, 4) SC with AATD AIMer (SA1 – 4). Liver biopsies day 7. RNA-seq to quantify on-target SERPINA1 editing, to quantify off-target editing reads mapped to entire mouse genome; plotted circles represent sites with LOD>3 (N=4), SERPINA1 edit site is indicated 19 Coverage Coverage

Proof of mechanism data in patients with AATD expected in 2024 Informs dose & RestorAATion-2: AATD Patients RestorAATion-1: Healthy Volunteers dose frequency SAD → MAD cohorts Dose E Up to 7 doses High dose Dose D Dose C Medium dose Dose B Low dose Multiple assessments of serum AAT throughout cohort Dose A Study key objectives Expect to initiate dosing in 4Q 2023 • Safety and tolerability• Serum M-AAT levels • Pharmacokinetics 20 HV: healthy volunteer; SAD: single-ascending dose; MAD: multi-ascending dose

AIMers RNA base editing capability

First-generation AIMer designs published in Nature Biotechnology • Specificity in vitro & in vivo (NHPs)• GalNAc conjugation • In vitro-in vivo translation (NHPs)• Foundational AIMer SAR AIMers detected in liver of NHP at Day 50 Substantial and durable editing in NHP ADAR editing with ACTB AIMer is (PK) liver in vivo highly specific (PD) RNA editing within full transcriptome (primary human hepatocytes) GalNAc AIMers Day 50 RNA editing ACTB in NHP GalNAc AIMers % Editing 22 RNA editing only detected at editing Monian et al., 2022 published online Mar 7, 2022; doi: 10.1038.s41587-022-01225-1 site in ACTB transcript SAR structure-activity relationship Confidence (LOD score)

Innovating on applications of ADAR editing Correct G-to-A driver mutations with AIMers Modulate protein interactions with AIMers Achieved POC Restore or correct Modulate protein- protein function protein interaction Upregulate expression WVE-006 Modify function (GalNAc AIMer) AATD Post-translational modification Alter folding or processing AIMers provide dexterity, with applications beyond precise correction of genetic mutations, including upregulation of expression, modification of protein function, or alter protein stability 23 POC: proof of concept

Proprietary base modifications increase editing across edit region sequences N3 U AIMer 5’ 3’ Edit Site Proprietary base modification (N3 U) increases UGP2 RNA editing across sequences in vitro ns ns * ** ** ** ** ** *** ** ** ** 100 • N3 U: example of proprietary base modifications 80 • N3 U consistently improves 60 RNA editing levels, including 40 across sequences 20 0 Seq 9 Seq 10 Seq 11 Seq 12 Seq 3 Seq 4 Seq 5 Seq 6 Seq 7 Seq 8 Seq 1 Seq 2 (GAA) (AAA) (CAC) (GAA) (CAG) (GAG) (GAU) (AAU) (CAC) (GAG) (GAA) (UAU) N3 U Cytosine 24 Presented at RNA Editing 2023 - Gordon Research Conference ADR-0106957 ADR-0106958 ADR-0106969 ADR-0106970 ADR-0106973 ADR-0106974 ADR-0106975 ADR-0106976 ADR-0106979 ADR-0106980 ADR-0106981 ADR-0106982 ADR-0106985 ADR-0106986 ADR-0106987 ADR-0106988 ADR-0106989 ADR-0106990 ADR-0106991 ADR-0106992 ADR-0106993 ADR-0106994 ADR-0106999 ADR-0107000 % Editing (mean ± sem) % Editing

Upregulation: AIMers can edit RNA motifs to restore or upregulate gene expression RNA binding proteins recognize sequence motifs to regulate mRNA stability Attenuated “Dialed up” Catalytically Efficient AIMer Gene Expression Gene Expression RNA-binding protein AIMer edits and durably stabilizes mRNA A mRNA Unique RNA motifs mRNA Decay Cascade Edited I(G) mRNA Decreased protein A single edited base production permanently disrupts Stable mRNA yields increased protein production the motif 25

Edit-verse subnetwork reveals “Target A”: Metabolic syndrome target uniquely suited for AIMer upregulation Target A • Liver target for upregulation, non-incretin therapy • Strongly implicated in metabolic disease, with indirect causation in familial disorders • Few therapies today provide weight loss in this specific patient population • Estimate 90 million potential patients in the US and Europe with metabolic syndrome and obesity • Serum protein levels and biomarkers available to assess target engagement 26 PoC: proof-of-concept Analysis of terminal endpoints (day 31) is shown. Each variable was analyzed using Welch’s two-sided t-test. Significance was evaluated a p<0.05.

Substantial upregulation of protein induces weight loss and improves insulin sensitivity • ~3-fold upregulation of Target A protein with GalNAc-AIMer led to weight reduction and improved insulin sensitivity in DIO mice Significant Weight Loss Improved Insulin Sensitivity Fasting glucose Fasting insulin (mg/dl) (ng/ml) AIMer Target A AIMer Target A p<0.05 * p<0.001 ** Body weight data were analyzed using a linear mixed effects model to assess the fix effects of diet, time and treatment, controlling for the initial day 0 body weight (continuous covariate) and subject (random effect). Fasted glucose and insulin data (from study termination, day 31) was analyzed using Welch’s two-sided t-test. Significance was evaluated 27 at p<0.05.

Target B upregulation offers a first-in-class therapeutic approach for hyperlipidemia >70% editing achieves ~2-fold upregulation with corresponding increase in protein Target B • Liver target for upregulation • Hyperlipidemia; first-in-class therapeutic approach • Estimate ~3 million target patients in US and Europe • Serum biomarkers available to assess target engagement and efficacy • Potential clinically meaningful benefit of >2 fold upregulation of target mRNA 28

Upregulation of liver Target X stops decline in kidney function Renal Insufficiency Target X Network • Liver target for upregulation • Target X produces a secreted protein to treat kidney disease • Estimate ~170K target patients in US and Europe • Therapeutic rationale supported by genetic insights, PheWAS, and observational data • Plasma biomarkers available to assess target engagement • ~2-fold upregulation in secreted protein expected to be clinically meaningful Target X 29

Building on success of AATD: Target E correction restores normal metabolism in rare genetic disease Proof-of-concept RNA editing in human primary Target E hepatocytes AIMer-1 Target E AIMer-2 Target E • Liver target for correction • Rare genetic disease • High unmet need population not addressed with current therapeutic options • ~17,000 patients addressable with correction approaches in US and Europe • Fully translatable serum biomarker • ~15-30% editing expected to deliver clinically p<0.001 * p<0.0001 ** meaningful benefit 30 Two-way ANOVA (Treatment, Dose) and Tukey’s HSD. Significance was evaluated at p<0.05, GalNAc-AIMers.

Upregulation of Target F restores kidney function in a rare genetic kidney disease Achieved >2-fold upregulation of Target F mRNA in vitro with RNA editing Target F • Kidney target for upregulation • Rare genetic kidney disease that leads to ESRD and need for dialysis / transplantation; High unmet need with few treatment options currently available • ~85K patients in US and Europe addressable with upregulation approach • Urinary biomarkers available to assess upregulation • Clinically meaningful benefit may be achieved with 2-fold upregulation ESRD: End Stage Renal Disease; Right: One-Way Anova; samples compared to NTC with Tukey’s HSD test. Significance evaluated at p<0.0001. 31

Correction of Target G mutation restores protein function in patients with a genetic lung disease Target G In vitro • Lung disease target for correction Target G mRNA editing 80 • Genetic lung disease with target patient population not addressed with available therapies 60 • ~5K patients amenable to correction approaches in 40 US and Europe 20 • Clinically meaningful benefit expected with 20% 0 correction • Established clinical regulatory pathway 32 Target G AIMER 1 Target G AIMer 2 Mock Percent Editing

Multiple RNA editing opportunities to build high-value pipeline beyond WVE-006 • The Edit-verse is substantial and still expanding • Advancing work for a diverse set of undisclosed targets addressing areas of high unmet need, including both rare and prevalent diseases Potential to advance any combination of targets into preclinical development Hepatic (GalNAc-AIMers) Extra-Hepatic (AIMers) Target A Target B Target X Target E Target F Target G Approach Upregulation Upregulation Upregulation Correction Upregulation Correction Tissue Liver Liver Liver Liver Kidney Lung Therapeutic Metabolic Metabolic Renal Rare Renal Rare Area Estimated Patients (US ~90M ~3M ~170K ~17K ~85K ~5K and Europe) 33

WVE-003 Huntington’s Disease

mHTT toxic effects lead to neurodegeneration; loss of wtHTT functions may also contribute to HD Huntington’s disease (HD) Healthy individual • Wild-type HTT (wtHTT) is critical for normal neuronal function • Expanded CAG triplet repeat in HTT gene results in production of mutant huntingtin protein (mHTT) wtHTT Stresses • HD is a monogenic autosomal dominant genetic disease; fully Huntington’s disease penetrant and affects entire brain • Fatal disease characterized by cognitive decline, psychiatric illness, and chorea ~50% decrease wtHTT Stresses mHTT + • 30,000 people with HD in the US in wtHTT and more than 200,000 at risk of Loss of wtHTT functions developing HD Synaptic dysfunction | Healthy CNS function Cell death | Neurodegeneration 35

WVE-003: First-in-class allele-selective candidate for HD Reductions in mean CSF mHTT and preservation of wtHTT observed in pooled analysis of single dose cohorts in SELECT-HD clinical study wtHTT protein levels mHTT protein levels Placebo WVE-003 (30 and 60 Reduction in mHTT mg pooled*) protein: 22% from baseline 35% vs. placebo Preservation of wtHTT Single dose Single dose of WVE-003 of WVE-003 Additional single-dose and available multi-dose data expected in 2H 2023 Complete multi-dose data from first cohort with extended follow-up expected 2Q 2024 mHTT: mutant huntingtin protein; wtHTT: wild-type huntingtin protein 36 *Pooled considering no apparent dose response between 2 cohorts; Data cut-off: August 29, 2022 Reductions in mHTT

INHBE and siRNA

Potential for best-in-class RNAi enabled by Wave’s PRISM platform • Unprecedented Ago2 loading increases potency and durability of silencing following administration of single subcutaneous dose Ago2 Ago2 ll ooa adindi g ng Antisense strand HSD17B13 mRNA (liver, transgenic mice) (liver, transgenic mice) (Liver, transgenic mice) 25 (liver, transgenic mice) 5 125 Wk 14 Wk 2 Wk 7 Wk 2 Wk 7 Wk 14 4 20 100 3 2 15 75 1 11 .0 1 50 10 0.5 **** * 25 * 5 0 0.0 0 2 4 6 8 10 12 14 16 0 Time (weeks) PBS Reference Wave siRNA Reference Wave siRNA RNAi is one of multiple Wave modalities being advanced in strategic research collaboration with GSK Left, Middle, and right: Mice expressing human HSD17B13 transgene treated with siRNA (3 mg/kg) or PBS, liver mRNA, guide strand concentration, Ago2 loading quantified. Stats: 38 Two-way ANOVA with post-hoc test * P<0.05, ****P<0.0001. Liu et al., 2023 Nuc Acids Res doi: 10.1093/nar/gkad268; % mRNA remaining (HSD17B13/Hprt) ug of oligo/g of tissue (mean± sem) Fold change relative to Reference 2

Driven by clinical genetics, Wave’s first RNAi program addresses high unmet need in metabolic disorders, including obesity INHBE program is Wave’s first wholly owned program emerging from GSK collaboration • Leverages novel genetic insights accessed through GSK collaboration 1,2,3 • INHBE loss-of-function heterozygous carriers exhibit healthy metabolic profile : ✓ Reduced waist-to-hip circumference ✓ Reduced odds ratio of Type 2 diabetes by 28%, and coronary artery disease ✓ Reduced serum triglycerides ✓ Elevated HDL-c ✓ Reduced HbA1c ✓ Lowered ApoB • INHBE expressed primarily in liver and gene product (subunit of activin E) acts on its 4 receptor in adipose tissue • GalNAc-siRNA for targeted delivery to hepatocytes ≥50% reduction of INHBE with siRNA expected to restore a healthy metabolic profile 1. Nat Commun 2022. https://doi.org/10.1038/s41467-022-32398-7; 2. Nat Commun 2022. https://doi.org/10.1038/s41467-022-31757-8; 3. PLOS ONE 2018. https://doi.org/10.1371/journal.pone.0194798; 4. Adam, RC. et.al. Proc Natl Acad Sci USA. 2023, 120(32): e2309967120. 39

INHBE GalNAc-siRNA represents an evolution in treatment for metabolic diseases, including obesity • Metabolic syndrome* is associated with type 2 diabetes, cardiovascular disease, hypertension, stroke, 1,2 cancer, and increased mortality • Estimate ~47M people in US and Europe with metabolic disorders, including obesity • Therapeutic options beyond GLP1s are needed 4 – GLP1 receptor agonists lead to weight loss at the expense of muscle 7 – GLP1 receptor agonists suppress general reward system 5 6 – GLP1 receptor agonists associated with poor tolerability profile with 68% drop-off after 1 year • Preferred approach would improve metabolism and increase fat loss while maintaining muscle mass • Restoration of metabolic health via INHBE silencing can simultaneously address obesity and other drivers of metabolic syndrome *Patients diagnosed with metabolic syndrome based on having 3 of the following: abdominal obesity, high bp, high blood glucose, high TG, or low HDL 1. Liang, et al. 2023 Postgraduate Medical Journal 99(1175):985; 2. Lakka, et al. 2002 JAMA 288(21):2709; 3. Ryan and Yockey 2017 Curr Obes Rep 6(2):187; 4. Sargeant, et al. 2019 Endocrinol Metab (Seoul) 34(3):247-262; 5. Liu, et al. 2022 Front. Endocrinol. 13:1043789; 6. Prime Therapeutics Claims Analysis, July 2023; 7. Müller, et al. 40 2019 Molecular Metabolism 30: 72-130.

INHBE knockdown of 90% demonstrated in human hepatocytes with GalNAc-siRNA Human hepatocytes Mouse hepatocytes 100 100 75 75 50 50 25 25 0 0 2 nM 8 nM 30 nM 125 nM 0.1 nM 1.6 nM 10 nM 25 nM • This cross-reactive sequence demonstrates ~90% maximal knock-down in human hepatocytes and ~65% in mouse hepatocytes • Additional human selective sequences are in development Primary hepatocytes were treated with a cross-reactive siRNA via free uptake. INHBE mRNA was quantified by RT-qPCR. 41 %mRNA remaining (SEM) (hINHBE/hSFRS9) %mRNA remaining (SEM) (mInhbe/mHprt)

INHBE silencing achieved in vivo with GalNAc-siRNA exceeds therapeutic threshold INHBE knockdown demonstrated in mice at 5 weeks ~62% 125 silencing 100 75 1 Therapeutic threshold **** 50 25 0 HFD: high-fat diet. Stats: two-sided Welch’s T Test **** P < 0.0001 42 1. Adam, RC. et.al. Proc Natl Acad Sci USA. 2023, 120(32): e2309967120. HFD, PBS Inhbe siRNA INHBE silencing mRNA expression (relative to PBS liver)

INHBE knockdown led to 16% lower body weight Similar effect seen in semaglutide preclinical studies Control Inhbe siRNA (HFD, PBS) 5 0 -5 -10 -15 -20 1 2 3 4 5 Weeks Data plotted by body weight difference as a percentage of PBS treated young DIO mice; Coskun, T. et. al. Mol. Metab. 2018, 18, 3. Stats: Repeated Measures ANOVA; Inhbe siRNA vs. Control significantly different at P < 0.05 level weeks 2 through 5 43 Lower relative body weight Body weight relative to PBS (%)

INHBE reduction leads to significant decrease in visceral fat at 5 weeks • INHBE knockdown in young DIO mice resulted in less fat mass across multiple types of white adipose tissue, without loss of brown fat Changes in white adipose tissue after 5 weeks inguinal mesenteric epididymal **** *** 1.5 1.5 ~34% reduction ~56% reduction *** 2.5 ~38% reduction 2.0 1.0 1.0 1.5 1.0 0.5 0.5 0.5 0.0 0.0 0.0 Stats: white-adjusted Two-way ANOVA with Bonferroni-adjusted post hoc comparisons per tissue type allowing heteroscedasticity (only HFD, Inhbe siRNA vs. HFD, PBS shown) *** P < 0.001, **** P < 0.0001 44 Chow, PBS HFD, PBS HFD, Inhbe siRNA Chow, PBS HFD, PBS HFD, Inhbe siRNA Chow, PBS HFD, PBS HFD, Inhbe siRNA mesWAT weight (g) epiWAT weight (g) ingWAT weight (g)

>50% reduction of INHBE mRNA recapitulates phenotype of heterozygous LoF carriers • Subsequent 8-week study demonstrates further reduction in excess visceral fat Changes in white adipose tissue after 8 weeks epididymal inguinal mesenteric **** ** ~40% reduction * ~56% reduction 1.5 2.5 1.5 ~45% reduction 2.0 1.0 1.0 1.5 1.0 0.5 0.5 0.5 0.0 0.0 0.0 First demonstration of siRNA treatment to restore healthy phenotype Adam, RC. et.al. Proc Natl Acad Sci USA. 2023, 120(32): e2309967120. HFD: high-fat diet. Stats: white-adjusted Two-way ANOVA with Bonferroni-adjusted post hoc 45 comparisons per tissue type allowing heteroscedasticity (only HFD, INHBE siRNA vs. HFD, PBS shown) * P < 0.05, ** P < 0.01, **** P < 0.0001 Chow, PBS HFD, PBS HFD, Inhbe siRNA Chow, PBS HFD, PBS HFD, Inhbe siRNA Chow, PBS HFD, PBS HFD, Inhbe siRNA mesWAT weight (g) epiWAT weight (g) ingWAT weight (g)

Wave’s next generation GalNAc-siRNA demonstrates best- in-class potential Next generation siRNA results in more potent and durable knockdown of serum Ttr protein 125 PBS • Applying next-generation Benchmark 100 siRNA chemistry to siRNA INHBE program 75 Next gen siRNA * • Potential for infrequent 50 * administration * 25 Next generation siRNA 0 0 5 10 15 20 25 30 35 40 45 50 Day INHBE candidate for metabolic disorders, including obesity, expected in 4Q 2024 Foster, DJ. et.al. Mol Ther. 2018, 26(3), 708. B6 mice administered PBS or 0.5 mg/kg of siRNA (subcutaneous). Benchmark: Stats: Mixed Two-way ANOVA followed by post hoc test comparing siRNA vs. Next gen siRNA per day derived from linear mixed effects model * P < 0.0001 46 Serum Ttr±SEM (rel to PBS)

Wave’s platform chemistry enables siRNA extra-hepatic delivery • Chemical impact – Introduction of neutral backbone – Unique structural feature of PN, specifically guanidine – Increased lipophilicity – Stereochemistry • Extra-hepatic delivery PN – Titrating siRNA lipophilicity tunable PNs (PN variants) – Maintaining high Ago2 loading and intracellular trafficking – Titrating plasma protein binding – Altered delivery, enhanced potency and durability in various tissues PN can tune extra-hepatic delivery of siRNA using rational design, including placement, number of modifications and PN variants 47

Tunable PN variants enhance potency and alter extra- hepatic delivery of non-GalNAc siRNAs Non-GalNAc siRNA with PN variants improve silencing in liver and adipose tissue 14 and 28 days post single dose • Reaching adipose tissue Day in addition to liver with siRNA is important for certain metabolic disorders *• PN variants also *** enhanced siRNA silencing in muscle **** tissue, including heart **** **** **** and diaphragm **** Stats: Three-way ANOVA followed by Bonferroni-adjusted post hoc test comparing condition to PBS (data not shown) * P < 0.05, *** P < 0.001, **** P < 0.0001; B6 mice administered PBS or 5 mg/kg of Sod1 siRNA (no GalNAc conjugate) subcutaneous injection (n=7). Taqman qPCR assays used for RNA PD, relative fold changes of Sod1 to Hprt 48 mRNA normalized to % of PBS group.

Single dose of next generation siRNA delivers broad, potent and durable CNS target engagement Sustained APP knockdown of at least 75% throughout the 16-week study in vivo in mice Next gen siRNA wk8 Next gen siRNA wk16 125 100 75 50 25 0 PBS (dotted line) or 100 μg of App siRNA administered ICV (n=7). PCR assays for RNA PD, relative fold changes of App to Hprt mRNA normalized to % of PBS; Stats: Three-way 49 ANOVA followed by Bonferroni-adjusted post hoc test comparing condition to PBS (data not shown), Next gen siRNA significantly lower than PBS at both time points for all tissues at P < 0.0001 level Cortex Striatum Cerebellum Hippocampus Brainstem Spinal cord APP silencing %mRNA remaining (SEM) (App/Hprt)

Wave siRNA demonstrates more potent and durable silencing as compared to published state-of-the-art Wave (APP – Cortex) Alnylam (APP – Cortex) Nat Biotechnol 40, 1500–1508 (2022) Single dose Single dose 120 μg by ICV 100 μg by ICV Next gen siRNA wk8 Next gen siRNA wk16 125 Knockdown < 90 days 100 post-dose Knockdown 75 > 112 days post-dose 50 25 0 Next gen N s eix R t N gA en w s ki8 RNA wk8 Next gen N s eix R t N gA en w s ki1 R6 NA wk16 125 125 PBS (dotted line) or 100 μg of App siRNA administered ICV (n=7). PCR assays for RNA PD, relative fold changes of App to Hprt mRNA normalized to % of PBS; Stats: Three-way ANOVA followed by Bonferroni-adjusted post hoc test comparing condition to PBS (data not shown), Next gen siRNA significantly lower than PBS at both time points for all tissues at P 50 < 0.0001 level. Source: Brown, K.M., Nair, J.K., Janas, M.M. et al. Expanding RNAi therapeutics to extrahepatic tissues with lipophilic conjugates. Nat Biotechnol 40, 1500–1508 100 100 (2022). 75 75 50 50 25 25 0 0 Cortex Striatum Cerebellum Cortex Hippocampus Cortex Striatum Brainstem Striatum Cerebellum Spinal cord Cerebellum Hippocampus Hippocampus Brainstem Brainstem Spinal cord Spinal cord %mRNA remaining (SEM) (App/Hprt) %mRNA remaining (SEM) (App/Hprt) %mRNA remaining (SEM) (App/Hprt)

Robust target engagement translates to substantial App protein reduction across brain regions Reductions observed 8-weeks post single-dose Frontal Cortex Striatum Hippocampus Cerebellum Brain Stem PBS Next gen siRNA Immunohistochemical analysis of FFPE Mouse Brain tissue labeling App protein (Color Brown) with CS#19389 followed by a ready to use Polymer-HRP nd 2 Detection antibody. Nuclei were counterstained with Hematoxylin (Color Blue). Single 100 ug ICV injection 51

First siRNA clinical candidate (INHBE) with proprietary chemistry expected in 4Q 2024 • INHBE GalNAc-siRNA program is driven by clinical genetics, with potential to be next- generation therapeutic for obesity – ≥50% silencing of INHBE is expected to improve metabolic health – INHBE siRNA silencing above therapeutic threshold restores healthy phenotype, with 16% lower body weight, as well as reduction of visceral fat to the level of lean-animals • Next generation GalNAc-siRNA formats are best-in-class and being applied to INHBE program • Wave’s platform chemistry enables extra-hepatic delivery for other non-hepatic targets – PN-variants on non-GalNAc siRNA enhance silencing in multiple tissues, including liver, adipose tissue and muscle. – Single dose of next generation siRNA delivers broad, potent (>75%) and durable CNS target engagement 52

Anticipated upcoming milestones

Anticipated upcoming milestones RNA ANTISENSE SPLICING RNAi EDITING SILENCING WVE-006 for AATD WVE-N531 for DMD WVE-003 for HD INHBE program for Most advanced RNA editing Potential best-in-class First-in-class metabolic disorders, candidate & potential best-in- approach with highest exon wild-type huntingtin protein including obesity class approach for AATD skipping reported (wtHTT)-sparing approach Driven by clinical genetics, with potential to be next- ✓WVE-006 CTA Additional single dose Dosing in potentially generation therapeutic for submissions in 2H 2023 and available multidose registrational obesity expected in 2H 2023 FORWARD-53 clinical Dosing in RestorAATion trial expected in 2023; Selection of INHBE clinical program in 4Q data expected in 2024 Complete multi-dose clinical candidate 2023; AAT protein data from first cohort of expected in 4Q 2024 restoration data expected SELECT-HD trial with Expansion opportunities in 2024 extended follow-up in other exons, as well Wave’s chemistry expected 2Q 2024 as other muscle and enables extra-hepatic Expansion opportunities in CNS diseases delivery for CNS and liver, CNS, and kidney Enables discussion on beyond next steps with Takeda DISCOVERY PIPELINE & COLLABORATIONS Advance collaboration activities with GSK, with potential for additional cash inflows in 2023 and beyond AATD: Alpha-1 antitrypsin deficiency; DMD: Duchenne muscular dystrophy; HD: Huntington’s disease 54

Wave is poised for significant and sustained growth INHBE Four additional metabolic AATD clinical disorders, WVE-006 candidates HD including obesity by year-end 2025 WVE-003 SNP3 Clinical candidate expected 4Q 2024 DMD WVE-N531 Add’l SNPs Exon 53 Add’l Exons ~$12B >$65B Value of US Total Addressable Market (TAM) 55 Note: Bubble size illustrative of size of total addressable US market (assuming 100% share of addressable patients)

For more information: InvestorRelations@wavelifesci.com 56
