UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: +
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading symbol |
Name of each exchange on which registered | ||
| Item 7.01 | Regulation FD Disclosure. |
From time to time, Wave Life Sciences Ltd. (the “Company”) presents and/or distributes slides and presentations to the investment community to provide updates and summaries of its business. On January 9, 2023, the Company shared an investor presentation, which is available on the “For Investors & Media” section of the Company’s website at http://ir.wavelifesciences.com/. This presentation is also furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The information in this Item 7.01 and exhibit 99.1attached hereto is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed incorporated by reference into any registration statement or other filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits.
| Exhibit No. | Description | |
| 99.1 | Investor Presentation of Wave Life Sciences Ltd. dated January 9, 2023 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| WAVE LIFE SCIENCES LTD. | ||
| By: | /s/ Paul B. Bolno, M.D. | |
| Paul B. Bolno, M.D. | ||
| President and Chief Executive Officer | ||
Date: January 9, 2023

Wave Life Sciences Corporate Presentation January 9, 2023 Exhibit 99.1

Forward-looking statements This document contains forward-looking statements. All statements other than statements of historical facts contained in this document, including statements regarding possible or assumed future results of operations, preclinical and clinical studies, business strategies, research and development plans, collaborations and partnerships, regulatory activities and timing thereof, competitive position, potential growth opportunities, use of proceeds and the effects of competition are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause the actual results, performance or achievements of Wave Life Sciences Ltd. (the “Company”) to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “aim,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this presentation are only predictions. The Company has based these forward-looking statements largely on its current expectations and projections about future events and financial trends that it believes may affect the Company’s business, financial condition and results of operations. These forward-looking statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties and assumptions, including those listed under Risk Factors in the Company’s Form 10-K and other filings with the SEC, some of which cannot be predicted or quantified and some of which are beyond the Company’s control. The events and circumstances reflected in the Company’s forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Moreover, the Company operates in a dynamic industry and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that the Company may face. Except as required by applicable law, the Company does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.

UNLOCKING THE BODY’S OWN ABILITY TO TREAT GENETIC DISEASE realizing a brighter future for patients and families

Building a leading genetic medicines company ALS: Amyotrophic lateral sclerosis; FTD: Frontotemporal dementia; HD: Huntington’s disease; DMD: Duchenne muscular dystrophy; AATD: Alpha-1 antitrypsin deficiency 1stereopure oligonucleotides and novel backbone chemistry modifications Diversified Pipeline CNS: ALS, FTD, HD Muscle: DMD Hepatic diseases: AATD Clinical Expertise Multiple global clinical trials Innovative trial designs Innovative Platform Stereopure oligonucleotides Novel backbone modifications (PN chemistry) Silencing, splicing, and editing modalities Strong and broad IP position1 GMP Manufacturing Internal manufacturing capable of producing oligonucleotides at scale LEVERAGING THE ONGOING genetic revolution Targeting THE TRANSCRIPTOME TO UNLOCK THE BODY’S OWN ABILITY TO TREAT GENETIC DISEASE >6,000 monogenic diseases; vastly more polygenic diseases Increase in genetic testing Biomarkers to assess target engagement early in clinical development Greater understanding of genetic disease and cellular biology Innovations for precise modification of transcriptome, proteome and interactome Many diseases out of reach for traditional medicines

… … Wave’s ability to rationally design oligonucleotides enables access to unique disease targets Chirality None PN backbone Sp PN backbone Rp Chirality … … PS backbone Rp PS backbone Sp Chirality … … PRISM backbone linkages PO: phosphodiester PS: phosphorothioate -O -S N (Rp) (Sp) PO PS PN Negative charge Neutral charge Negative charge Phosphoryl guanidine x-ray structure example

Harnessing the biological machinery in our cells to treat genetic diseases Silencing Splicing RNA Base Editing Degradation of RNA transcripts to turn off protein production Restore RNA transcripts and turn on protein production Efficient editing of RNA bases to restore or modulate protein production Endogenous ADAR enzyme Restored Reading Frame Endogenous RNase H Endogenous AGO2 RISC Functional Protein Skip

Built-for-Purpose Candidates to Optimally Address Disease Biology Silencing | Splicing | RNA Editing DESIGN Unique ability to construct stereopure oligonucleotides and control three structural features to efficiently engage biological machinery OPTIMIZE Provides the resolution to observe this structural interplay and understand how it impacts key pharmacological properties Sequence Stereochemistry Chemistry Unlocking the body’s own ability to treat genetic disease

THERAPEUTIC AREA / TARGET MODALITY DISCOVERY PRECLINICAL CLINICAL RIGHTS NEUROLOGY ALS and FTD C9orf72 Takeda 50:50 option Huntington’s disease mHTT SNP3 SCA3 ATXN3 CNS diseases Multiple 100% Global DMD Exon 53 HEPATIC AATD – lung and liver disease SERPINA1 GSK Exclusive Global License Robust portfolio of stereopure, PN-modified oligonucleotides ALS: Amyotrophic lateral sclerosis; FTD: Frontotemporal dementia; SCA3: Spinocerebellar ataxia 3; CNS: Central nervous system; DMD: Duchenne muscular dystrophy; AATD: Alpha-1 antitrypsin deficiency Therapeutic modality Silencing Splicing ADAR editing (AIMers) WVE-004 (FOCUS-C9) WVE-003 (SELECT-HD) WVE-006 WVE-N531 NEUROLOGY HEPATIC (GalNAc)

GSK Collaboration and WVE-006 for Alpha-1 antitrypsin deficiency (AATD)

Collaboration leverages Wave’s unique stereopure, PN-chemistry containing PRISM™ platform, including editing, splicing, silencing (RNAi and antisense) Strategic collaboration with GSK to develop transformative RNA therapeutics for genetically defined diseases 1$120 million in cash and $50 million equity investment, 2Initiation, development, launch, and commercialization milestones for programs progressed during initial 4-year research term (WVE-006 and 8 GSK collaboration programs) 3GSK eligible to receive tiered royalty payments and commercial milestones from Wave First-in-class RNA editing program GSK receives exclusive global license to WVE-006 for AATD GSK to advance up to eight collaboration programs Up to $225 million in development and launch milestones Up to $1.2 billion in aggregate in initiation, development and launch milestones Up to $300 million in sales-related milestones Up to $1.6 billion in aggregate in sales-related milestones Double-digit tiered royalties as a percentage of net sales up to high-teens Tiered royalties as a percentage of net sales up to low-teens Development and commercialization responsibilities transfer to GSK after completion of first-in-patient study Development and commercialization responsibilities transfer to GSK at development candidate Wave to advance up to three wholly owned collaboration programs (or more pending agreement with GSK) 3 Wave to leverage GSK’s genetically-validated targets Multiple value drivers to Wave Milestone / royalties Genetic targets Milestone / royalties $170 million upfront to Wave (cash and equity1) Additional research support funding Potential for up to $3.3 billion in milestones2 Expands Wave’s pipeline Extends cash runway into 2025

3) Retain M-AAT physiological regulation 2) Reduce Z-AAT protein aggregation in liver WVE-006: Designed to correct mutant SERPINA1 transcript to address both liver and lung manifestations of AATD M-AAT reaches lungs to protect from proteases M-AAT secretion into bloodstream AAT: Alpha-1 antitrypsin Strnad et al., 2020 N Engl J Med 382:1443-55; Blanco et al., 2017 Int J Chron Obstruct Pulmon Dis 12:561-69; Remih et al., 2021 Curr Opin Pharmacol 59:149-56. WVE-006 ADAR editing approach to address key goals of AATD treatment: RNA correction replaces mutant Z-AAT protein with wild-type M-AAT protein Z-AAT 1) Restore circulating, functional wild-type M-AAT I(G) A SERPINA1 Z allele mRNA encodes Z-AAT protein with E342K mutation Edited SERPINA1 mRNA enables wild-type M-AAT protein production WVE-006 (GalNAc-conjugated AIMer) WVE-006 designed to correct Z allele mRNA to enable M-AAT protein to be produced

~50% RNA editing expected to increase PI*ZZ patient serum AAT levels to PI*MZ levels, with low risk of disease 1. Brode, et al. 2012 CMAJ 184:1365-1371; 2. ATS/ERS. 2003 Am J Respir Crit Care Med 168:818–900. Serum AAT Protein Levels and Risk of AATD by Genotype Risk of Emphysema Very High High Low Very Low No Risk of Liver Disease No High Possible Possible No ~50% RNA editing ~2.5-7-fold higher 100% Z-AAT protein 100% M-AAT protein

WVE-006 supports dose-dependent RNA editing in human preclinical model systems Left: MZ-donor derived primary human hepatocytes treated with WVE-006 at indicated concentrations for 48 hours Right: Patient-iPSC derived hepatocytes (ZZ genotype) plated on day 0 and treated on day 2 with WVE-006 at indicated concentrations. Media refreshed every 2 days (days 4, 6, 8). RNA was collected on day 10. In each experiment, RNA editing was quantified by Sanger sequencing (n=2 biological replicates) % Editing (mean, sem) iPSC-derived human hepatocytes (ZZ genotype) 0.31 1.25 5.0 0.08 Concentration (mM) Note: Due to MZ genotype, Y-axis ranges from ~50-100% Efficient SERPINA1 editing in donor-derived primary human hepatocytes with WVE-006 (MZ genotype) 0 2 4 6 8 10 Days: Dose Collect RNA Dose Refresh media

WVE-006 results in circulating AAT protein levels 7-fold above PBS control, well above established 11µM threshold WVE-006 administered subcutaneously (10 mg/kg bi-weekly) in 7-week old NSG-PiZ mice (n=5 per group); Loading dose: 3 x 10 mg/kg at Day 0. Left: Liver biopsies collected at week 13 (one week after last dose) and SERPINA1 editing was quantified by Sanger sequencing; Stats: One-way ANOVA with adjustment for multiple comparisons (Tukey); Right: Total serum AAT protein quantified by ELISA; Stats: Two-Way ANOVA with adjustment for multiple comparisons (Tukey) SERPINA1 mRNA editing in liver of AATD mouse model (NSG-PiZ mice) (Week 13) SERPINA1 editing WVE-006 treatment results in serum AAT protein levels >11 uM in AATD mouse model (NSG-PiZ mice) Restored AAT protein
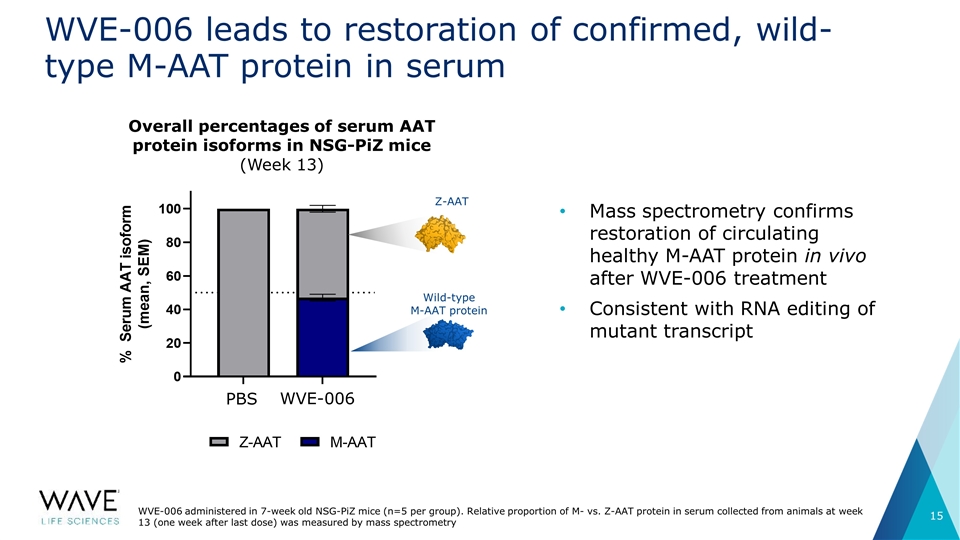
WVE-006 leads to restoration of confirmed, wild-type M-AAT protein in serum WVE-006 administered in 7-week old NSG-PiZ mice (n=5 per group). Relative proportion of M- vs. Z-AAT protein in serum collected from animals at week 13 (one week after last dose) was measured by mass spectrometry Overall percentages of serum AAT protein isoforms in NSG-PiZ mice (Week 13) PBS WVE-006 Wild-type M-AAT protein Mass spectrometry confirms restoration of circulating healthy M-AAT protein in vivo after WVE-006 treatment Consistent with RNA editing of mutant transcript Z-AAT

Significant increase in neutrophil elastase inhibition activity indicates restored M-AAT protein is functional GalNAc-conjugated AIMers administered in 7-week old NSG-PiZ mice (n=5 per group). Serum collected from mice was tested for ability to inhibit fixed concentration of neutrophil elastase in an in vitro reaction. Stats: Two-way ANOVA with adjustment for multiple comparisons (Bonferroni) >3-fold increase over PBS PBS WVE-006 Increased neutrophil elastase inhibition activity demonstrates functionality of AAT protein Increases in neutrophil elastase, a proteolytic enzyme, may cause emphysema and damage the surrounding lung tissue Main function of AAT protein is to neutralize/control neutrophil elastase Serum neutrophil elastase inhibition activity

Early lead pre-optimization AATD AIMer (SA1-5) administered in huADAR/SERPINA1 mice (8–10 wKs old); lower left: 20x liver images PAS-D stained, 19 weeks; Quantification of PAS-D positive staining, Stats 2-way ANOVA; Right: Quantification lobular inflammation grade (Grade based on # of inflammatory foci in lobules: Grade 0: 0; G1 1-5; G2 6-10; G3 11-15; G4 ≥16) and mean globular diameter (40 largest globules/ animal) with HALO. Stats Wilcox rank-sum tests Early lead (pre-optimization) AATD AIMer reduces aggregation of Z-AAT and inflammation in mouse liver Lobular inflammation (19 weeks) * p=0.03 Inflammation grade PBS AIMer p<0.01 Mean diameter (mm) PBS AIMer **** p<0.0001 Weeks following first dose ** %PAS-D positive area (mean±sem) PBS Early lead AATD AIMer

RNA editing only detected at PiZ mutation site in SERPINA1 transcript (mouse liver) RNA editing across transcriptome (mouse liver) AIMer-directed editing is highly specific in mice SERPINA1 (PiZ mutation site) % Editing Dose 3x10 mg/kg (days 0, 2, 4) SC with AATD AIMer (SA1 – 4). Liver biopsies day 7. RNA-seq to quantify on-target SERPINA1 editing, to quantify off-target editing reads mapped to entire mouse genome; plotted circles represent sites with LOD>3 (N=4), SERPINA1 edit site is indicated No bystander editing observed on SERPINA1 transcript Coverage Coverage Editing site (PiZ mutation) PBS AATD AIMer C 0% T 100% C 48.2% T 51.8%

WVE-006 is a potential first- and best-in-class candidate for AATD Correct Z-allele mRNA to replace mutant Z-AAT protein with functional wild-type M-AAT protein RNA editing levels show potential to support conversion of a patient from ZZ to MZ mRNA expression M-AAT protein can address lung disease Reduction of Z-AAT protein enables clearance of protein aggregates in liver M-AAT protein produced with WVE-006 would remain under physiological regulation mRNA editing is highly specific Potentially applicable across AATD patient subpopulations Convenience of subcutaneous administration
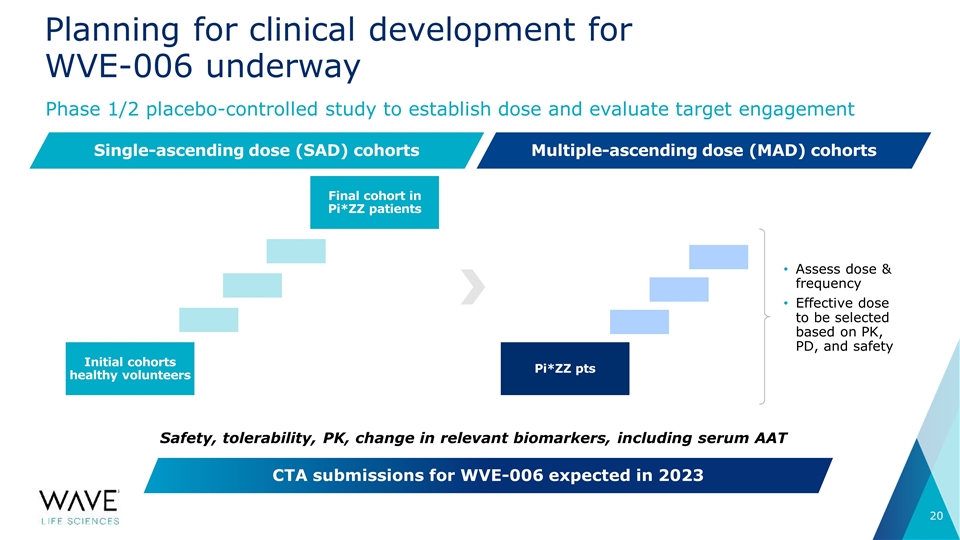
Planning for clinical development for WVE-006 underway Phase 1/2 placebo-controlled study to establish dose and evaluate target engagement CTA submissions for WVE-006 expected in 2023 Single-ascending dose (SAD) cohorts Multiple-ascending dose (MAD) cohorts Initial cohorts healthy volunteers Final cohort in Pi*ZZ patients Pi*ZZ pts Assess dose & frequency Effective dose to be selected based on PK, PD, and safety Safety, tolerability, PK, change in relevant biomarkers, including serum AAT

WVE-N531 Duchenne muscular dystrophy

Duchenne muscular dystrophy Genetic mutation in dystrophin gene prevents the production of dystrophin protein, a critical component of healthy muscle function Impacts approx. 1 in every 5,000 newborn boys each year; approx. 20,000 new cases annually worldwide Approx. 8-10% are amenable to exon 53 skipping Dystrophin protein established by FDA as surrogate endpoint reasonably likely to predict benefit in boys1 for accelerated approval in DMD Increasing amount of functional dystrophin expression over minimal amount shown with approved therapies is expected to result in greater benefit for boys with DMD 1Vyondys: www.fda.gov; viltepso; www.fda.gov; Exondys; www.fda.gov; Amondys: www.fda.gov Dysfunctional Splicing Exon Skipping No dystrophin protein produced Functional dystrophin produced Translation halted Translation continues Mutant pre-mRNA Disease State Restored State mRNA with disrupted reading frame Restored mRNA Mutant pre-mRNA Skip Oligo 53 53 50 51 54 55 50 51 54 55 53 50 51 54 55 50 51 54 55

PN chemistry improved muscle exposure and survival in preclinical mouse models Kandasamy et al., 2022; doi: 10.1093/nar/gkac018 PN increased muscle concentrations after single dose, which correlated with exon-skipping activity PN PN Treatment with PN-modified molecules led to 100% survival of dKO mice at time of study termination Better tissue exposure 100 75 50 25 0 Survival probability (%) 0 4 8 12 16 20 24 28 32 36 40 Time (weeks) PS/PO/PN, 75 mg/kg bi-weekly PBS PS/PO, 150 mg/kg weekly PS/PO/PN, 150 mg/kg weekly Note: Untreated, age-matched mdx mice had 100% survival at study termination [not shown]

PS/PO/PN splicing compound restores muscle and respiratory function to wild-type levels in dKO mice Left: Mdx/utr-/- mice received weekly subQ 150 mg/kg dose of PS/PO/PN stereopure oligonucleotide (postnatal day 10). Age-matched mdx/utr-/- littermates treated with PBS, wild-type C57BL10 mice not treated. Wild-type, dKO PBS mice: 6 wks old; dKO PS/PO/PN: 28 – 41 wks old; Electrophysiology performed at Oxford University based on Goyenvalle et al., 2010 Mol Therapy; Right: Kandasamy et al., 2022; doi: 10.1093/nar/gkac018 Wild-type dKO / PBS dKO (PS/PO/PN oligonucleotide) **** **** **** **** Specific Force (EDL) Eccentric Concentration dKO: PS/PO/PN dKO: PBS Wild-type Muscle Function Respiratory Function

WVE-N531: Dystrophin restoration in vitro and enhanced muscle distribution in NHPs Western Blot normalized to primary healthy human myoblast lysate Dystrophin protein restoration of up to 71% in vitro Plasma and tissue concentrations of WVE-N531 (PS/PO/PN) significantly higher than suvodirsen (first-generation PS/PO) WVE-N531 concentrations in heart and diaphragm substantially higher than skeletal muscle concentrations Higher plasma Cmax, AUC and Ctrough Enhanced muscle distribution in NHPs Preclinical data supported advancing proof-of-concept study to rapidly assess impact of PN chemistry in splicing oligonucleotides

In multidose portion of study, patients received three biweekly 10 mg/kg doses Initial cohort Boys with DMD amenable to exon 53 skipping 1 mg/kg 3 mg/kg 6 mg/kg 10 mg/kg Single ascending intra-patient doses Multidosing at 10 mg/kg every other week Weeks Dose WVE-N531 Period before initiating multidosing (~1 – 2 months) 10 mg/kg 10 mg/kg 10 mg/kg 0 2 4 6 Muscle Biopsy Data include: WVE-N531 muscle concentrations WVE-N531 localization Exon skipping Dystrophin protein

WVE-N531 appeared safe and well-tolerated All treatment-emergent adverse events (TEAEs) were mild, except one COVID-19 infection of moderate intensity All adverse events (AEs) related to study drug (headache, pruritic rash) were mild, transient and resolved without sequelae No serious adverse events (SAEs) No events met stopping criteria No trend for an increase in TEAEs with single dose escalation from 1 to 10 mg/kg or three repeat doses at 10 mg/kg No evidence of class-related risks, such as thrombocytopenia, coagulation, complement activation, cytokine activation

Plasma pharmacokinetic profile enabling meaningful WVE-N531 tissue concentrations Plasma concentrations and other PK parameters following a single dose of 10 mg/kg demonstrate a half-life of 25 days Higher plasma concentrations For 10 mg/kg dose level: Cmax: 191 (+/- 18.1) (µg/mL) AUClast : 933 (+/- 103) (µg*h/mL) Ctrough : 53 (+/- 10) (ng/mL) t1/2 : 25 days

Mag: 40x with an enlarged images Myofibers WVE-N531 (in red) in myofiber cytoplasm (stars) and nuclei (yellow arrows) RNAscope (ISH – in situ hybridization) Control Probes: Ubiquitin – Positive DapB – Negative Intracellular WVE-N531 enabling PD effects

High muscle concentration and exon skipping indicate WVE-N531 is engaging target Mean exon skipping: 53% Mean muscle concentration: 42 µg/g Biopsies collected ~2 weeks post-last dose (3 biweekly doses of 10 mg/kg) 42 µg/g = 6.1 µM BLQ: Below level of quantification (1%) Patient Tissue Source Tissue concentration (µg/g) % Exon skipping by RT-PCR Dystrophin by Western blot (% of normal) 1 Deltoid 85.5 61.5 0.24 2 Deltoid 33.5 49.8 0.23 3 Bicep 8.3 47.9 0.34 Mean dystrophin: 0.27% of normal (BLQ)

Conclusions & next steps Achieved proof-of-concept: High muscle concentrations of WVE-N531 and exon skipping observed following three biweekly doses at 10 mg/kg Planning underway to continue to evaluate dystrophin Evaluating next steps for program in light of evolving regulatory environment

WVE-004 Amyotrophic Lateral Sclerosis (ALS) Frontotemporal Dementia (FTD)

C9orf72 repeat expansions: One of the most common genetic causes of ALS and FTD Typically 100’s-1000’s of GGGGCC repeats Amyotrophic Lateral Sclerosis (ALS) Frontotemporal Dementia (FTD) Hexanucleotide (G4C2)- repeat expansions in C9orf72 gene are common autosomal dominate cause for ALS and FTD Different manifestations across a clinical spectrum Fatal neurodegenerative disease Progressive degeneration of motor neurons in brain and spinal cord C9-specific ALS: ~2,000 patients in US Progressive neuronal degeneration in frontal / temporal cortices Personality and behavioral changes, gradual impairment of language skills C9-specific FTD: ~10,000 patients in US Including patients with C9-associated ALS, FTD or both Sources: Balendra et al, EMBO Mol Med, 2017; Brown et al, NEJM, 2017, DeJesus-Hernandez et al, Neuron, 2011. Renton et al, Neuron, 2011. Zhu et al, Nature Neuroscience, May 2020, Stevens et al, Neurology 1998

Reduced expression RNA variants RAN translation C9orf72 protein RNA foci Dipeptide repeat proteins (DPRs) Sense: poly(GA), poly(GR) Antisense: poly(PR), poly(PA) Sense & Antisense: poly(GP) Toxic RNA aggregation Gain-of-function Loss-of-function Repeat-expanded allele Wild-type C9orf72 allele Genetic mutation C9orf72 Poly(GP) biomarker selected as preferred DPR biomarker Abundant in CNS Most soluble Stable expression Only DPR derived from both sense & antisense RNAs Variant-selective oligonucleotide, lowering V1 & V3 in preclinical studies1 Preserves C9orf72 protein expression; does not exacerbate potential loss-of-function driver of disease Reduces toxic gain-of-function drivers of disease (RNA foci, DPRs) 1Liu et al., 2022 Mol Ther Nuc Acids doi: 10.1016/j.omtn.2022.04.007 Transcription Antisense Mis-spliced RNA Stabilized intron 1 Pathological RNAs V1 V2 V3 WVE-004 is designed to affect multiple drivers of toxicity Disease drivers Sense & Antisense RNA Decrease in beneficial protein WVE-004 addresses each biological aspect of C9orf72-associated ALS and FTD 1 2 3

* *** ** *** Spinal cord Relative poly(GP) levels (normalized to PBS) Cortex >90% knockdown of poly(GP) DPR protein Two doses of WVE-004 Six months >80% knockdown of poly(GP) DPR protein Relative poly(GP) levels (normalized to PBS) p≤0.0001 Liu et al., 2022 Molecular Therapy Nucleic Acids doi: 10.1016/j.omtn.2022.04.007; 2 x 50 ug (day 0, day 7) dosed ICV; DPRs measured by poly(GP) MSD assay. *: p≤ 0.05 **: P ≤ 0.01, ***: P ≤ 0.001. DPR: Dipeptide repeat protein Weeks Weeks PBS Poly(GP) DPR Oligonucleotide concentration WVE-004: WVE-004: C9orf72 protein unchanged at 6 months ns ug of oligo / g of tissue ug of oligo / g of tissue ns Relative fold change C9orf72/HPRT1 1.5 0.5 0.0 1.0 Relative fold change C9orf72/HPRT1 1.5 0.5 0.0 1.0 WVE-004 PBS WVE-004 PBS Preclinical studies with WVE-004 demonstrated durable reduction of poly(GP) in spinal cord and cortex 6 months after two doses Preclinical in vivo results: Spinal cord Cortex

WVE-004 clinical data demonstrate successful translation of preclinical approach to clinic PK: pharmacokinetic PD: pharmacodynamic; Right: Mixed model for repeated measures used to estimate geometric mean ratio to baseline via least squares mean and to calculate p-values. P-values represented by asterisks are for within-dose group geometric mean ratios. *p≤0.05, **p≤0.01, ***p≤0.001. Poly(GP) assay: Wilson et al., 2022 J Neurol Neurosurg Psychiatry doi:10.1136/jnnp-2021-328710. Data presented at ENCALS Meeting (June 1-3, 2022) and International Congress on Frontotemporal Dementias (Nov. 2 – 5, 2022) PK/PD modeling using preclinical in vivo models predicted pharmacodynamically active starting dose Target engagement confirmed in patients supports advancing FOCUS-C9 clinical study Poly(GP) reduction in cortex and spinal cord in transgenic mice with WVE-004 Sufficient concentrations of WVE-004 in cortex and spinal cord of NHP for target engagement

30 mg n=10 Dosing ongoing in FOCUS-C9 clinical trial with multiple doses of WVE-004 10 mg n=3 60 mg n=4 20 mg n=10 10 mg n=6 4 monthly doses Dose and frequency to be guided by DSMB Target engagement observed in single dose cohorts Single dose Multidose OLE clinical trial initiated in 4Q 2022 10 mg n=10 Quarterly doses 20 mg n=10 Quarterly doses Open-label extension (OLE) Data from all cohorts in the FOCUS-C9 trial are expected in 1H 2023

WVE-003 Huntington’s Disease

Healthy individual Huntington’s disease mHTT toxic effects lead to neurodegeneration, loss of wtHTT functions may also contribute to HD Stresses wtHTT Stresses wtHTT mHTT + ~50% decrease in wtHTT Healthy CNS function Synaptic dysfunction | Cell death | Neurodegeneration Loss of wtHTT functions Huntington’s disease (HD) Wild-type HTT (wtHTT) is critical for normal neuronal function* Expanded CAG triplet repeat in HTT gene results in production of mutant huntingtin protein (mHTT) HD is a monogenic autosomal dominant genetic disease; fully penetrant and affects entire brain Fatal disease characterized by cognitive decline, psychiatric illness, and chorea 30,000 people with HD in the US and more than 200,000 at risk of developing HD

mHTT, mutant HTT; wtHTT, wild-type HTT; PO, phosphodiester; PS, phosphorothioate; PN, phosphoryl guanidine; wtHTT literature sources: 1. Leavitt 2006 2. Cattaneo 2005 3. Kumar 2016 4. Franco-Iborra 2020 5. Hamilton 2015 6. Ochaba 2014 7. Wong 2014 8. Rui 2015 9. Caviston 2007 10. Twelvetrees 2010 11. Strehlow 2007 12. Milnerwood 2010 13. Smith-Dijak 2019 14. Tousley 2019 15. Zhang 2018 16. McAdam 2020 17. Altar 1997 18. Zuccato 2001 19. Gauthier 2004 20. Ferrer 2000 21. Baquet 2004 22. Liu 2011 23. Karam 2015 wtHTT supports healthy brain function, especially in the context of stress Promotes neuronal survival Supports synaptic protein transport Regulates synaptic plasticity Supports cilia and CSF circulation Unique and innovative wildtype HTT-sparing oligonucleotide WVE-003: Only investigational HD therapy in clinical development designed to lower mHTT while sparing wtHTT Delivered to CNS without invasive surgical procedures WVE-003 Designed with next-generation PN chemistry No complex delivery vehicles required (e.g. AAV)

Hu97/18 mice administered 3x100 mg intracerebroventricular doses PBS or oligonucleotide. Relative mHTT RNA in cortex (left) striatum (middle) or hippocampus (right) at 4, 8 and 12-weeks post-dosing. Data are mean ± SD, n=8. Stats: ns non-significant, *P<0.05, **P<0.01, ***P<0.0001, ****P<0.0001 versus PBS by 1-way ANOVA. mHTT, mutant HTT; wtHTT, wild-type HTT; Tubb, tubulin Allele-selective molecule decreases mHTT, spares wtHTT; Pan-silencer uniformly decreases both Percentage HTT RNA expression (mHTT/Tubb - PBS) **** *** **** **** **** mHTT wtHTT Hu97/18 mouse Allele-selective activity in CNS of Hu97/18 mice **** * *** Cortex Striatum PBS Pan-silencing control Allele-selective molecule Time (weeks) Time (weeks) PBS Pan-silencing control Allele-selective molecule

WVE-003 (SNP3) demonstrates selective, potent, and durable reduction of mHTT in preclinical models Selectively reduces mHTT mRNA in HD iPSC neurons in vitro Results from ND50036 iPSC-derived medium spiny neurons. Total HTT knockdown quantified by qPCR and normalized to HPRT1. Oligonucleotide or PBS [100 μg ICV injections through cannula on days 1, 3, 5] delivered to BACHD transgenic. Mean ± SD (n=8, *P<0.0332, ***P<0.0002, ****P<0.0001 versus PBS unless otherwise noted). HPRT1, hypoxanthine-guanine phosphoribosyl transferase; iPSC, induced pluripotent stem cell; ICV, intracerebroventricular; PBS, phosphate-buffered saline Similar results in cortex Pan-silencing reference compound WVE-003 PBS Weeks *** **** **** **** **** **** Pan-silencing reference compound WVE-003 Percentage HTT mRNA Remaining Durable striatal mHTT knockdown for 12 weeks in BACHD mouse model

Initial clinical results indicating allele-selective target engagement suggests translation of preclinical data Allele selectivity (Hu97/18 mice) mHTT reduction in cortex and striatum (transgenic mice) Concentrations in NHP brain tissues sufficient for target engagement PK/PD modeling using preclinical in vivo models mHTT: mutant huntingtin proteinwtHTT: wild-type huntingtin protein*Pooled considering no apparent dose response between 2 cohorts mHTT protein levels Placebo WVE-003 (30 and 60 mg pooled*) wtHTT protein levels Reductions in mean CSF mHTT and preservation of wtHTT observed in pooled analysis of single dose cohorts in SELECT-HD clinical study Single dose of WVE-003 or placebo Single dose of WVE-003 or placebo

Expanding single dose cohorts to optimize dose level based on initial clinical results 30 mg Expanding cohort 90 mg Expanding cohort Additional single-dose biomarker and safety data are expected in 1H 2023 mHTT protein reductions observed in single dose cohorts wtHTT protein levels appear consistent with allele-selectivity Generally safe and well-tolerated Adapting clinical trial to optimize dose level 60 mg Expanding cohort mHTT: mutant huntingtinwtHTT: wild-type huntingtin Adding patients to each cohort

AIMers RNA base editing capability

Unlocking RNA editing with PRISM platform to develop AIMers: A-to-I editing oligonucleotides Endogenous ADAR enzymes 1Woolf et al., PNAS Vol. 92, pp. 8298-8302, 1995; Right: Data from independent experiments; Total RNA was harvested, reverse transcribed to generate cDNA, and the editing target site was amplified by PCR and quantified by Sanger sequencing Improved editing PS/PO/PN PS/PO (Stereopure) PS/PO (Stereorandom) Concentration (mM) % ACTB editing ADAR enzymes First publication (1995) using oligonucleotide to edit RNA with endogenous ADAR1 Catalyze conversion of A-to-I (G) in double-stranded RNA substrates A-to-I (G) edits are one of the most common post-transcriptional modifications ADAR1 is ubiquitously expressed across tissues, including liver and CNS Learnings from biological concepts Applied to ASO structural concepts Applied PRISM chemistry AIMer: Wave’s A-to-I editing oligonucleotides Free-uptake of chemically modified oligonucleotides (No need for LNPs or viral vectors) Stereochemistry and PN chemistry enhance potency and editing efficiency of GalNAc AIMers in primary human hepatocytes

AIMers: Realizing potential of therapeutic RNA editing by harnessing endogenous ADAR Solved for key therapeutic attributes for potential best-in-class RNA editing therapeutics Efficient ADAR recruitment AIMer design principles SAR developed to design AIMers for different targets Systematized AIMer design enables rapid advancement of new targets Strong and broad IP in chemical and backbone modifications, stereochemistry patterns, novel and proprietary nucleosides Potent and specific editing in vivo Efficient ADAR recruitment Stability Delivery and intracellular trafficking Beyond liver Decade of investment and learnings to improve stability of single-stranded RNAs GalNAc compatible for targeted liver delivery Endosomal escape and nuclear uptake AIMer design also works for delivery to CNS and other tissue types Potential for infrequent dosing Subcutaneous dosing IT, IVT, systemic dosing SAR: structure-activity relationship

Proof-of-concept preclinical RNA editing data published in Nature Biotechnology (March 2022) Monian et al., 2022 published online Mar 7, 2022; doi: 10.1038.s41587-022-01225-1 SAR structure-activity relationship Specificity in vitro & in vivo (NHPs) In vitro-in vivo translation (NHPs) GalNAc conjugation Foundational AIMer SAR AIMers detected in liver of NHP at Day 50 (PK) ADAR editing with ACTB AIMer is highly specific ACTB Confidence (LOD score) % Editing RNA editing within full transcriptome (primary human hepatocytes) Substantial and durable editing in NHP liver in vivo (PD) Day 50 RNA editing in NHP RNA editing only detected at editing site in ACTB transcript GalNAc AIMers GalNAc AIMers

Editing: Potent, durable, specific A à I (G) RNA editing Delivery: Efficient RNA editing in preclinical in vivo models: Targeted delivery (GalNAc) Systemic delivery Local delivery (IT, IVT, others) Potential to accelerate timelines to candidate with AIMer pipeline expansion Systemic in vivo editing without delivery vehicles Right: Single dose of 100mg/kg unconjugated UGP2 AIMer, seven days post dose; WAT: White adipose tissue; BAT: Brown adipose tissue; CD3+: T-cells and subset of NK cells; EpCAM+(Epithelial cell adhesion molecule): mainly cholangiocytes within liver; LSEC cells (Liver Sinusoidal Endothelial Cells); M0 cells: macrophages Substantial RNA editing across multiple tissues following single subcutaneous dose of UGP2 AIMer Specific liver associated cells Editing without GalNAc conjugation Control UGP2 AIMer (unconjugated) T cell NK cell subset Cholan-giocyte LSEC Macro-phage

Substantial in vivo editing without delivery vehicles in CNS tissues Transgenic huADAR mice administered 100 mg AIMer or PBS on day 0 and evaluated for UGP2 editing across CNS tissues at 1, 4, 8, 12, and 16-weeks post dose. Percentage UGP2 editing determined by Sanger sequencing. Stats: 2-way ANOVA compared to PBS (n=5 per time point per treatment) *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001. ICV intracerebroventricular; PBS phosphate buffered saline Peak RNA editing observed one-month post-single dose across tissues 30% >40% 25% >40% 50% >65% Peak editing UGP2 AIMer-1 PBS Potential CNS editing targets to benefit from learnings taken from clinical CNS silencing programs

Expanding addressable disease target space using AIMers to activate pathways and upregulate expression Modulate protein-protein interaction Upregulate expression Modify function Post-translational modification Alter folding or processing Restore or correct protein function Achieved POC WVE-006 (GalNAc AIMer) AATD POC: proof of concept Correct G-to-A driver mutations with AIMers Modulate protein interactions with AIMers AIMers provide dexterity, with applications beyond precise correction of genetic mutations, including upregulation of expression, modification of protein function, or alter protein stability

n=2; Primary hepatocytes 48h of treatment with the indicated dose concentration of AIMers NRF2 is degraded by proteasome NRF2 KEAP1 Transcription is repressed ADAR editing site NRF2 is stabilized KEAP1 NRF2 Transcription is activated Basal conditions ADAR-modified conditions AIMer RNA editing efficiency Dose-dependent gene upregulation (NQO1) in vitro following Nrf2 editing to disrupt protein/protein interaction Gene upregulation NRF2 AIMers UGP2 (Control) AIMer Dose dependent modulation of protein/protein interactions

Note: Editing percentage for UGP2 control AIMer indicates editing of UGP2 mRNA Methods: hADAR C57BL/6 mice dosed subQ (days 0, 2, 4) at 10mg/kg GalNAc-conjugated AIMers. Livers harvested (day 7), analyzed for editing and NQO1 expression via Sanger sequencing or qPCR, respectively. Data analyzed via One-way ANOVA with Tukey’s multiple comparison test. Asterisks indicate statistical significance to PBS-treated animals as follows: * = p<0.05; ** = p<0.01; *** = p<0.001; **** = p<0.0001 AIMers enable activation of gene pathway in vivo with single edit AIMer 1 UGP2 AIMer AIMer 2 RNA editing efficiency Gene upregulation NRF2 downstream gene upregulation following GalNAc AIMer mRNA editing in vivo in liver of mice RNAseq transcriptome analysis confirms disruption of Nrf2 protein interaction with upregulation of key factors Nrf2 mRNA editing in vivo in liver of mice with GalNAc AIMers

Upregulation: AIMers can edit RNA motifs to restore or upregulate gene expression RNA binding proteins recognize sequence motifs to regulate various mRNA properties Enhance or inhibit mRNA decay Stability Intracellular localization Transport Splicing Capping Processing PolyA usage Translational efficiency Protein production mRNA AIMer edits mRNA à “dials up” gene expression A I(G) Decay No binding RNA binding protein Edited mRNA
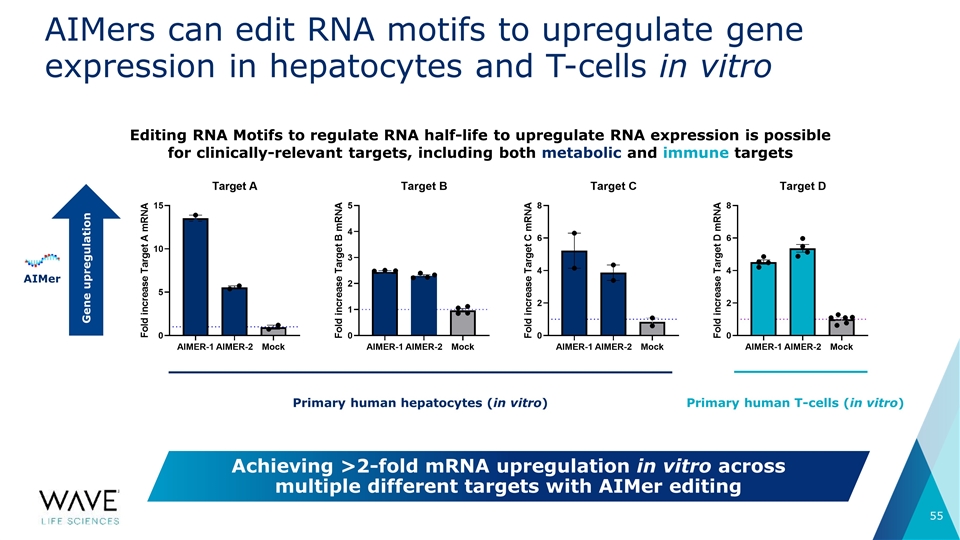
AIMers can edit RNA motifs to upregulate gene expression in hepatocytes and T-cells in vitro Editing RNA Motifs to regulate RNA half-life to upregulate RNA expression is possible for clinically-relevant targets, including both metabolic and immune targets Gene upregulation Primary human hepatocytes (in vitro) Primary human T-cells (in vitro) AIMer Achieving >2-fold mRNA upregulation in vitro across multiple different targets with AIMer editing

Proof-of-concept: Considerations to translate Target A upregulation results in vivo Gene upregulation AIMer Response is greater in human hepatocytes ~14-fold compared to mouse hepatocytes ~2-fold In vitro Target A mRNA fold upregulation (gymnotic 10uM, ~48h) Target A (undisclosed liver target) High unmet need with potential for multiple large indications Preserves endogenous protein function Serum protein with biomarkers of pathway activation Potential benefit 3-fold+ upregulation in mouse models Human Mouse Mouse model may underpredict potential translation of Target A upregulation

AIMers upregulate mRNA and downstream serum protein in vivo above anticipated threshold In vitro to in vivo translation of mouse Target A mRNA upregulation In vivo mRNA upregulation corresponds to an upregulation of Target A protein in serum at Day 7 demonstrating proof-of-concept mRNA upregulation 7 days post-initial dose GalNAc AIMer Protein upregulation 7 days post-initial dose GalNAc AIMer Upregulation mRNA editing 7 days post-initial dose GalNAc AIMer Target A (undisclosed liver target) High unmet need with potential for multiple large indications Preserves endogenous protein function Serum protein with biomarkers of pathway activation Potential benefit 3-fold+ upregulation in mouse Potential threshold for benefit hADAR mouse dosed subcutaneously 3 x 10 mg/kg GalNAc-conjugated AIMer or PBS days (0, 2, 4), taken down at day 7 RNA editing

RNA editing of nonsense mutation found in MECP2 (Rett Syndrome) restores functional protein 293T cells transfected with both nonsense mutation on MECP2 (GFP-fusion construct) and ADAR plasmids. AIMers transfected for 48h prior to RNA extraction and sequencing. Percentage editing determined by Sanger sequencing. Left: Single dose (25nM) treatment Middle: Full dose response curve (25nM, 5-fold dilution, 48h treatment) in presence or absence of hADAR Right: Western blot for MECP2 protein. Three biological replicates, NTC AIMer, mock and naïve 293T cells probed for fusion protein. in vitro ADAR editing of over 60% targeting MECP2 disease transcript Full length MECP2 protein is expressed following ADAR editing Loading Control Endogenous MECP2 ADAR Edited MECP2 Mock Naive NTC Ladder Dose-dependent RNA editing of MECP2 mutation with PS/PN AIMer Control (no hADAR) PS/PN AIMer … CGA… wild type protein … TGA… premature stop codon … TGG… restored protein Normal: Rett Syndrome: ADAR editing: Variant base ADAR editing site Nonsense mutations found in Rett Syndrome can occur in multiple locations on RNA transcript: PN chemistry improved editing efficiency in vitro Dosed with hADAR

Wave’s discovery and drug development platform

Improvements in PRISMTM primary screen hit rates accelerate drug discovery over time Primary screen hit rates with silencing far above industry standard hit rates Stereorandom Chemistry, PN stereochemistry & machine learning optimization Stereopure Chemistry improvements and PRISM advancement All screens used iPSC-derived neurons; Data pipeline for improved standardization. Hit rate = % of oligonucleotides with target knockdown greater than 50%. Each screen contains >100 oligonucleotides. ML: machine learning (2019) (2020 - current)

Silencing Potency is enhanced with addition of PN modifications across modalities Improved knockdown Splicing Editing Improved skipping Ranked by potency of reference PS/PO compound Ranked by potency of reference PS/PO compound Improved editing PS/PO/PN PS/PO (Stereopure) PS/PO (Stereorandom) Concentration (mM) % Editing PS/PO reference compound PS/PN modified compound % Skipping Target knockdown (% remaining) Left: Experiment was performed in iPSC-derived neurons in vitro; target mRNA levels were monitored using qPCR against a control gene (HPRT1) using a linear model equivalent of the DDCt method; Middle: DMD patient-derived myoblasts treated with PS/PO or PS/PO/PN stereopure oligonucleotide under free-uptake conditions. Exon-skipping efficiency evaluated by qPCR. Right: Data from independent experiments

Adding PN chemistry modifications to C9orf72-targeting oligonucleotides improved potency in vivo Exposure (µg/g) Exposure (µg/g) Cortex %C9orf72 V3 transcript remaining Oligonucleotide concentrations quantified by hybridization ELISA. Graphs show robust best fit lines with 95% confidence intervals (shading) for PK-PD analysis; Liu et al. Molecular Therapy Nucleic Acids 2022; Kandasamy et al., Nucleic Acids Research, 2022, doi: 10.1093/nar/gkac037 Spinal Cord C9orf72-targeting oligonucleotides PS/PO backbone chemistry PS/PO/PN backbone chemistry Improved knockdown Improved tissue exposure

PN chemistry improves distribution to CNS NHPs administered 1x12 mg oligonucleotide or PBS by intrathecal injection/lumbar puncture (IT). CNS tissue evaluated 11 or 29 days after injection (n=6 per group). Oligonucleotide was visualized by ViewRNA (red), and nuclei are counterstained with hematoxylin. Images from day 29. Cerebral Cortex Cerebellum Striatum Hippocampus Spinal cord PS/PO PS/PO/PN Backbone chemistry Midbrain Distribution of oligonucleotides in non-human primate CNS 1-month post single IT dose Oligonucleotide (red staining)

PRISMTM PN siRNA led to unprecedented silencing >3 months after single dose PBS HSD Reference 2 HSD PN/PS/PO Wk2 Wk 14 Wk 7 Reference 2 HSD PN/PS/PO **** * * Mice expressing a human HSD17B13 transgene were treated with 3 mg/kg of the indicated siRNA or PBS, and liver mRNA, guide strand concentration, and Ago2 loading were quantified at the indicated times post-dose. Stats: Two-way ANOVA with post-hoc test * P<0.05, ****P<0.0001. Reference 2 is based on Foster, et al., 2018. Mol. Ther. 26, 708-717 Ago2 loading (liver, transgenic mice)

Established internal GMP manufacturing for multiple oligonucleotide modalities Strong technical knowhow and operating expertise Established infrastructure State of the art facilities (90,000 sq ft) and expansion space Process and analytical development labs GMP oligonucleotide (API) manufacturing Established Quality and GMP systems (QA, supply chain, logistics, QC testing) Experienced team led by Sridhar Vaddeboina, PhD (SVP Chemistry, Manufacturing, Controls) Experts in oligonucleotide synthesis (ASOs, DNAs, RNAs, siRNAs) Proven track record scaling complex chemistries; delivered clinical supply for six programs at Wave Scalable to support Wave’s GMP manufacturing needs, as well as potential new partners

Upcoming Milestones

Data updates inform future opportunities, unlock value and cash Silencing CNS (Intrathecal) Splicing Muscle (IV) Correction Targeted delivery to liver (Subcutaneous) Multiple modalities Opportunities in a variety of tissues and delivery mechanisms WVE-004 C9orf72 ALS & FTD Delivered clinical target engagement data with single doses Initiated OLE clinical trial in 4Q 2022 Data from all cohorts in FOCUS-C9 trial expected in 1H 2023 WVE-003 HD SNP3 Delivered single-dose clinical data indicating reduction in mHTT with wtHTT preserved, appearing consistent with allele-selectivity Additional single-dose biomarker and safety data in 1H 2023 WVE-N531 DMD Exon 53 Achieved proof-of-concept based on high muscle concentrations and exon skipping Planning underway to continue to evaluate dystrophin WVE-006 AATD Selected an AATD AIMer development candidate and initiated IND-enabling activities Submit clinical trial applications in 2023 Collabor-ations Entered into collaboration with GSK – multiple value drivers including adding up to 3 Wave programs with novel targets & up to $3.3B in milestones for programs initiated in next 4 years WVE-004 FOCUS-C9 clinical trial (NCT04931862); WVE-003 SELECT-HD clinical trial (NCT05032196) WVE-N531 open-label clinical trial (NCT04906460) Cash runway into 2025

Realizing a brighter future for people affected by genetic diseases For more information: Kate Rausch, Investor Relations InvestorRelations@wavelifesci.com 617.949.4827
